This event has ended. Keep an eye on our website and social media for more fun events!
Hillman Hall of Minerals and Gems
New Mineral Acquisitions
I had a successful trip to Tucson, Arizona in January/February of this year. This is an annual event where the Section of Minerals participates in the Tucson Gem & Mineral Show by not only exhibiting a mineral display, the Carnegie Mineralogical Award is also presented during the show, and many of the minerals acquired for the collection are found amongst the dozens of venues around the city where vendors are selling their specimens. Numerous motels turned into shopping centers starting as early as January 28th, where each room is a separate store for an individual vendor. Tent shows were set up along streets and in parking lots. The Tucson Convention Center housed two major shows: The American Gem Trade Association Show (AGTA), which was held February 5th through 10th, and the Tucson Gem & Mineral Show, which was held February 14th through 17th. In total, I brought back 10 specimens acquired for the collection, five of which were acquired specifically for exhibit in Hillman Hall of Minerals and Gems or Wertz Gallery: Gems and Jewelry. Two gemstones were put on display in the “What is a Gemstone?” exhibit in Wertz Gallery on March 12th: a 44.27 carat, cushion cut spodumene from Afghanistan, and a 7.08 carat, trillion cut titanite (also known as sphene) from Zimbabwe.


A special exhibit to highlight the museums acquisitions will be put in Hillman Hall on March 26th that will feature a world class Kermesite specimen from China. This specimen measures 20 cm and is probably the finest example of its species in the world. The largest kermesite in our collection prior to this acquisition is only 3cm.

Two other specimens will be going on display soon in the Systematic Collection area of Hillman Hall: a blue tabular beryl from Afghanistan in the Silicates 2 case, and a bornite from Montana in the Sulfides 2 case. The beryl is a recent discovery in Afghanistan that is different than any other type of beryl, while the bornite was collected sometime in the 1950s in Butte, Montana which is known as the best locality in the United States for this species. Watch for announcements of when these two special pieces go on exhibit!


Debra Wilson is the Collection Manager for the Section of Minerals at Carnegie Museum of Natural History. Museum employees are encouraged to blog about their unique experiences and knowledge gained from working at the museum.
How Do Geodes Get Their Colors?

Geodes are stones with a secret–at first glance, they seem like nothing more than the grey rocks you might see on walk around your neighborhood. Then–crack!–once they’re broken open, they reveal their inner beauty: a tiny cave filled with some of the world’s most spectacular, colorful crystals.
Born out of lava
Geodes are formed when there are pockets of air within rocks. This often happens after volcanic eruptions when lava cools around air bubbles. These pockets leave space for groundwater to seep in. But the water itself doesn’t produce geodes–it brings along minerals which stay in the rock even after the water evaporates. The minerals then start to build on each other to form crystals. It can take thousands or even millions of years for these crystals to form. The larger the crystals are, the older the geode is.
So what gives them their color?

The same minerals that form crystals can give them their glorious colors. Additional elements can also make their way into the mix and provide their own unique shades. Iron will give crystals a red or purple color, titanium will create blue, nickel or chromium leads to green, and manganese produces pink crystals.
While geodes can be naturally colorful some are artificially dyed. These dyed stones often have a brighter, more intense color than what appears naturally. Why do people dye geodes? Colorful geodes tend to sell well and can be a cheap way to imitate rare stones.
Come to the museum and check out the geodes of various colors on display in Hillman Hall of Minerals and Gems.
Explore nature together. Visit Nature 360 for more activities and information.
A Perfect Mineral for the Christmas Season
by Debra Wilson
How can you decorate a mineral for Christmas? If it’s the right mineral, all you need to do is shine a SWUV (short wave ultraviolet) light on it. Such is the case with a mineral that is known as “Christmas Ore.” Under normal light it looks like kind of a drab rock as the one in this photo does.

This specimen of calcite (tan color), willemite (brown color) and franklinite (black color) is on display in Hillman Hall of Minerals and Gems in the Fluorescence & Phosphorescence exhibit and was donated by the Sterling Hill Mining Museum for this exhibit. It originated from the Sterling Hill mine in Ogdensburg, in the Franklin Mining District of New Jersey. When you shine the SWUV light on it you will see why it is called “Christmas Ore” because it glows with the colors of Christmas. The calcite glows a bright red and the willemite glows a bright green, as you can see in this photo.

This glowing is known as fluorescence and the Franklin Mining District is known as the fluorescent capital of the world. The Franklin and Sterling Hill ore bodies are the source of at least 350 mineral species.At present, over 80 fluorescent mineral species are known from the area. Willemite and calcite are the most common fluorescents in these ore bodies and are known as “Christmas Ore” when they occur together in the same specimen.
So, what causes the fluorescence? Fluorescence usually occurs when specific impurities known as “activators” are present within the mineral. These activators are typically cat ions of metals such as: tungsten, molybdenum, lead, boron, titanium, manganese, uranium and chromium. Rare earth elements such as europium, terbium, dysprosium, and yttrium are also known to contribute to the fluorescence phenomenon. Fluorescence can also be caused by crystal structural defects or organic impurities. Calcite (CaCO3) and willemite (Zn2SiO4) are examples of minerals that in their pure state do not fluoresce but add a little divalent manganese (Mn2+) and they will fluoresce red and green, respectively.
There are two other specimens of calcite and willemite, also from the Franklin District, in the Fluorescence & Phosphorescence exhibit, shown here under normal light and under SWUV light.




Come to Hillman Hall of Minerals and Gems to hear a more detailed explanation of the phenomenon of fluorescence and see all 21 specimens in the exhibit from world-wide localities that glow under the ultraviolet lights.
Debra Wilson is the Collection Manager for the Section of Minerals at Carnegie Museum of Natural History. Museum employees are encouraged to blog about their unique experiences and knowledge gained from working at the museum.
Related Content
Thanksgiving and Nutritional Mineralogy
Ask a Scientist: Why do some minerals glow?
Carnegie Museum of Natural History Blog Citation Information
Blog author: Wilson, DebraPublication date: December 13, 2018
Share this post!
Fred the Crystal Skull
by Debra Wilson
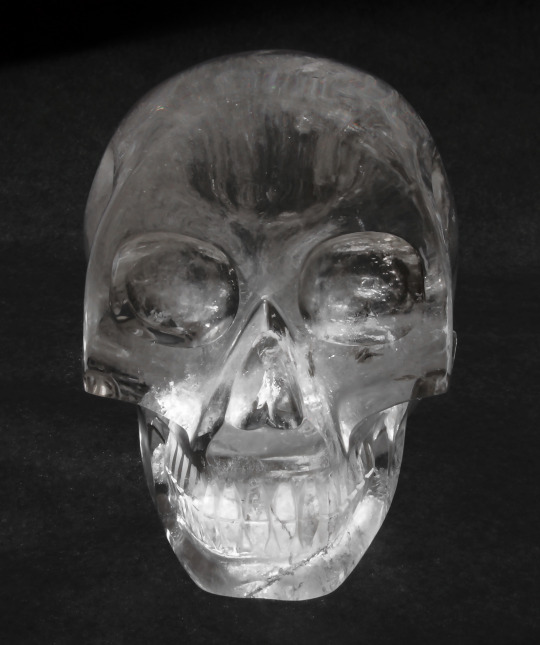
Just about every year since the Carnegie Museum of Natural History acquired it, Fred the Crystal Skull has made an appearance in Hillman Hall of Minerals and Gems right around Halloween. So how did we acquire a crystal skull and how did it get the name Fred you ask? Just to set the record straight right off the bat, Fred is not one of the dozen or so mysterious skulls that some think were carved by an ancient Mesoamerican civilization thousands of years ago. Our skull was carved and polished from a single quartz crystal with modern tools in Brazil and was donated to the museum in 2004 by South American Gems, Ltd located in Guarapari, Espirito Santo, Brazil.Germany, China and Brazil currently produce thousands of carved crystal skulls every year in numerous sizes. Fred measures 7.8 inches high by 5 inches wide, which is slightly smaller than the average human skull (8 to 9 inches high and 6 to 7 inches wide) so he was named after a man of small stature, namely the step father of the former Head of the Section of Minerals Marc Wilson. Marc was Section Head from August 1992 to August 2017.

As you can see in the photograph of Fred, he has some internal flaws and fractures which is very common in the mineral quartz. Chemical impurities, physical flaws and twinning in natural quartz are issues that caused industry to develop a commercial process of manufacturing pure, electronics-grade quartz that can be used in circuits for consumer products such as televisions, radios, computers, cell phones and electronic games, just to name a few, and for crystal-controlled clocks and watches. As it so happens, the Section of Minerals also has a few lab-grown quartz crystals in the collection, including a large crystal nicknamed The Football that is nearly a foot across.

You will notice it is so clear that you can see the growth patterns of the bottom surface through the crystal. The Football was part of a donation of 57 lab created specimens given to the Section of Minerals in 2017 by Lynn Boatner just before he retired from Oak Ridge National Laboratory in Tennessee.
Debra Wilson is the Collection Manager for the Section of Minerals at Carnegie Museum of Natural History. Museum employees are encouraged to blog about their unique experiences and knowledge gained from working at the museum.
Related Content
BBQ Chips

How about some BBQ chips with your burger?
Just kidding! BBQ chips is the nickname for this wulfenite with mimetite specimen on display in the Masterpiece Gallery.
